Further progress in the understanding of atoms did not occur until the science of chemistry began to develop. In 1661, natural philosopher Robert Boyle published The Sceptical Chymist in which he argued that matter was composed of various combinations of different "corpuscules" or atoms, rather than the classical elements of air, earth, fire and water.[13] In 1789 the term element was defined by the French nobleman and scientific researcher Antoine Lavoisier to mean basic substances that could not be further broken down by the methods of chemistry.[14]
In 1803, English instructor and natural philosopher John Dalton used the concept of atoms to explain why elements always react in a ratio of small whole numbers—the law of multiple proportions—and why certain gases dissolve better in water than others. He proposed that each element consists of atoms of a single, unique type, and that these atoms can join together to form chemical compounds.[15][16] Dalton is considered the originator of modern atomic theory.[17]
Additional validation of particle theory (and by extension atomic theory) occurred in 1827 when botanist Robert Brown used a microscope to look at dust grains floating in water and discovered that they moved about erratically—a phenomenon that became known as "Brownian motion". J. Desaulx suggested in 1877 that the phenomenon was caused by the thermal motion of water molecules, and in 1905 Albert Einstein produced the first mathematical analysis of the motion.[18][19][20] French physicist Jean Perrin used Einstein's work to experimentally determine the mass and dimensions of atoms, thereby conclusively verifying Dalton's atomic theory.[21]

In 1869, building upon earlier discoveries by such scientists as Lavoisier, Dmitri Mendeleev published the first functional periodic table.[22] The table itself is a visual representation of the periodic law which states certain chemical properties of elements repeat periodically when arranged by atomic number.
Subcomponents and quantum theory
The physicist J. J. Thomson, through his work on cathode rays in 1897, discovered the electron, and concluded that they were a component of every atom. Thus he overturned the belief that atoms are the indivisible, ultimate particles of matter.[23] Thomson postulated that the low mass, negatively-charged electrons were distributed throughout the atom, possibly rotating in rings, with their charge balanced by the presence of a uniform sea of positive charge. This later became known as the plum pudding model.
In 1909, Hans Geiger and Ernest Marsden, under the direction of physicist Ernest Rutherford, bombarded a sheet of gold foil with alpha rays—by then known to be positively charged helium atoms—and discovered that a small percentage of these particles were deflected through much larger angles than was predicted using Thomson's proposal. Rutherford interpreted the gold foil experiment as suggesting that the positive charge of a heavy gold atom and most of its mass was concentrated in a nucleus at the center of the atom—the Rutherford model.[24]
While experimenting with the products of radioactive decay, in 1913 radiochemist Frederick Soddy discovered that there appeared to be more than one type of atom at each position on the periodic table.[25] The term isotope was coined by Margaret Todd as a suitable name for different atoms that belong to the same element. J.J. Thomson created a technique for separating atom types through his work on ionized gases, which subsequently led to the discovery of stable isotopes.[26]
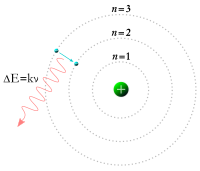
Meanwhile, in 1913, physicist Niels Bohr suggested that the electrons were confined into clearly defined, quantized orbits, and could jump between these, but could not freely spiral inward or outward in intermediate states.[27] An electron must absorb or emit specific amounts of energy to transition between these fixed orbits. When the light from a heated material was passed through a prism, it produced a multi-colored spectrum. The appearance of fixed lines in this spectrum was successfully explained by these orbital transitions.[28]
Chemical bonds between atoms were now explained, by Gilbert Newton Lewis in 1916, as the interactions between their constituent electrons.[29] As the chemical properties of the elements were known to largely repeat themselves according to the periodic law,[30] in 1919 the American chemist Irving Langmuir suggested that this could be explained if the electrons in an atom were connected or clustered in some manner. Groups of electrons were thought to occupy a set of electron shells about the nucleus.[31]
In 1918, Rutherford discovered that the positive charge within every atom was always equal to an integer multiple of hydrogen nuclei, and deduced that all nuclei contained positively-charged particles called protons. The mass of the nucleus often exceeded this multiple, however, and speculated that the excess mass to be composed of neutrally-charged particles (neutrons).[citation needed]
The Stern–Gerlach experiment of 1922 provided further evidence of the quantum nature of the atom. When a beam of silver atoms was passed through a specially shaped magnetic field, the beam was split based on the direction of an atom's angular momentum, or spin. As this direction is random, the beam could be expected to spread into a line. Instead, the beam was split into two parts, depending on whether the atomic spin was oriented up or down.[32]
In 1926, Erwin Schrödinger, using Louis de Broglie's 1924 proposal that particles behave to an extent like waves, developed a mathematical model of the atom that described the electrons as three-dimensional waveforms, rather than point particles. A consequence of using waveforms to describe particles is that it is mathematically impossible to obtain precise values for both the position and momentum of a particle at the same time; this became known as the uncertainty principle, formulated by Werner Heisenberg in 1926. In this concept, for a given accuracy in measuring a position one could only obtain a range of probable values for momentum, and vice versa. This model was able to explain observations of atomic behavior that previous models could not, such as certain structural and spectral patterns of atoms larger than hydrogen. Thus, the planetary model of the atom was discarded in favor of one that described atomic orbital zones around the nucleus where a given electron is most likely to be observed.[33][34]

The development of the mass spectrometer allowed the exact mass of atoms to be measured. The device uses a magnet to bend the trajectory of a beam of ions, and the amount of deflection is determined by the ratio of an atom's mass to its charge. The chemist Francis William Aston used this instrument to show that isotopes had different masses. The atomic mass of these isotopes varied by integer amounts, called the whole number rule.[35] The explanation for these different isotopes awaited the discovery of the neutron, a neutral-charged particle with a mass similar to the proton, by the physicist James Chadwick in 1932. Isotopes were then explained as elements with the same number of protons, but different numbers of neutrons within the nucleus.[36]
Fission, high energy physics and condensed matter
In 1938, the German chemist Otto Hahn, a student of Rutherford, directed neutrons onto uranium atoms expecting to get transuranium elements. Instead, his chemical experiments showed barium as a product.[37] A year later, Lise Meitner and her nephew Otto Frisch verified that Hahn's result were the first experimental nuclear fission.[38][39] In 1944, Hahn received the Nobel prize in chemistry in which, despite the efforts of Hahn, the contributions of Meitner and Frisch were not recognized.[40]
In the 1950s, the development of improved particle accelerators and particle detectors allowed scientists to study the impacts of atoms moving at high energies.[41] Neutrons and protons were found to be hadrons, or composites of smaller particles called quarks. Standard models of nuclear physics were developed that successfully explained the properties of the nucleus in terms of these sub-atomic particles and the forces that govern their interactions.[42]
Around 1985, Steven Chu and co-workers at Bell Labs developed a technique for lowering the temperatures of atoms using lasers. In the same year, a team led by William D. Phillips managed to contain atoms of sodium in a magnetic trap. The combination of these two techniques and a method based on the Doppler effect, developed by Claude Cohen-Tannoudji and his group, allows small numbers of atoms to be cooled to several microkelvin. This allows the atoms to be studied with great precision, and later led to the Nobel prize-winning discovery of Bose-Einstein condensation.[43]
Historically, single atoms have been prohibitively small for scientific applications. Recently, devices have been constructed that use a single metal atom connected through organic ligands to construct a single electron transistor.[44] Experiments have been carried out by trapping and slowing single atoms using laser cooling in a cavity to gain a better physical understanding of matter.



0 comments:
Post a Comment